Max Delbrück Center for Molecular Medicine
←
→
Transkription von Seiteninhalten
Wenn Ihr Browser die Seite nicht korrekt rendert, bitte, lesen Sie den Inhalt der Seite unten
https://www.mdc-berlin.de/de/veroeffentlichungstypen/clinical- journal-club Als gemeinsame Einrichtung von MDC und Charité fördert das Experimental and Clinical Research Center die Zusammenarbeit zwischen Grundlagenwissenschaftlern und klinischen Forschern. Hier werden neue Ansätze für Diagnose, Prävention und Therapie von Herz-Kreislauf- und Stoffwechselerkrankungen, Krebs sowie neurologischen Erkrankungen entwickelt und zeitnah am Patienten eingesetzt. Sie sind eingelanden, um uns beizutreten. Bewerben Sie sich!
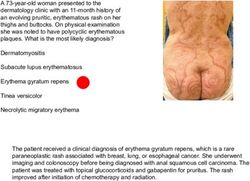
A baby boy born at 39 weeks of gestation had bilious
emesis, failure to pass meconium, and abdominal
distention within 24 hours after birth. A water-soluble
contrast enema showed a uniformly distended and
shortened colon. What is the most likely diagnosis?
Hirschsprung’s disease
Congenital syphilis
Pyloric stenosis
Duodenal atresia
Necrotizing enterocolitis
The correct answer is Hirschsprung’s disease. The
patient was taken to the operating room where biopsy
specimens of the colon revealed the absence of
ganglion cells. Abundant ganglion cells were found at
the level of the terminal ileum, and an end ileostomy
was performed followed by colectomy with stapled side-
to-side anastomosis of the terminal ileum next to the
rectal remnant 6 months later.Beim kongenitalen Megakolon (Synonym Megacolon congenitum, angeborenes Megakolon, aganglionotisches Megakolon, Hirschsprung-Krankheit, Morbus Hirschsprung) handelt es sich um eine angeborene Erkrankung des Dickdarms aus der Gruppe der Aganglionosen. Dabei fehlen in einem Abschnitt des Dickdarms Nervenzellen, wodurch sich die dortige Muskulatur zusammenzieht und den Darm verengt. Der Stuhl staut sich vor der Engstelle, wodurch sich der davorliegende Teil des Darms erweitert (dilatiert). Systematischer Erstbeschreiber (1886) ist der dänische Pädiater Harald Hirschsprung (1830–1916). Im Durchschnitt tritt diese Fehlbildung bei 1 von 5000 Kindern auf, wobei Jungen im Vergleich deutlich häufiger betroffen sind als Mädchen. Bei ca. 12 % der Säuglinge mit einem Down-Syndrom (Trisomie 21) ist Morbus Hirschsprung nachweisbar. Kombinationen mit anderen Fehlbildungen (z. B. Mukoviszidose, Brachydaktylie) kommen vor, sind aber selten. In 80 % der Fälle sind bei der Aganglionose nur das Rektum und/oder Sigma betroffen (Short-Segment- Aganglionose). Etwa 5 % gehören zur Long-Segment- Aganglionose, bei der der krankhaft veränderte Dickdarmabschnitt insgesamt 40 cm und mehr ausgedehnt ist. In weniger als 5 % der Fälle fehlen die Nervenzellen im gesamten Abschnitt des Dickdarmes, man spricht in diesem Fall von einem Jirásek-Zuelzer- Wilson-Syndrom. In einigen Fällen fehlen die Nervenzellen bis in den Dünndarm. Das kongenitale Megakolon kann auch im Rahmen von Syndromen auftreten, siehe Mowat-Wilson-Syndrom.
Ein Mangel an Ganglienzellen (Aganglionose) im Bereich des Plexus submucosus (Meißner-Plexus) bzw. myentericus (Auerbach-Plexus) führt zu einer Hyperplasie (übermäßige Zellbildung) der vorgeschalteten parasympathischen Nervenfasern mit vermehrter Acetylcholin-Ausschüttung. Durch diese permanente Stimulation der Ringmuskulatur kommt es zu einem dauerhaften Zusammenziehen des betroffenen Darmabschnittes. Das übermäßig gebildete Acetylcholin wird durch eine als Ausgleich vermehrt produzierte Acetylcholinesterase abgebaut. Späte Defekte der Neuroblasteneinwanderung, Reifungsstörungen eingewanderter Neuroblasten, zeitweilige Ischämien (Minderdurchblutung) des Darms oder virale Infektionen beim Embryo kommen als Ursache in Frage. Bei Untersuchungen zum Morbus Hirschsprung wurden Mutationen im sogenannten Ret-Protoonkogen (autosomal dominante Form), im Endothelin-3-Gen (EDN3) und im Endothelinrezeptor-Gen (EDNRB) (autosomal rezessive Form) belegt. Among the patients in our study, Hirschsprung's disease arose from common noncoding variants, rare coding variants, and copy-number variants affecting genes involved in enteric neural-crest cell fate that exacerbate the widespread genetic susceptibility associated with RET. For individual patients, the genotype-specific odds ratios varied by a factor of approximately 67, which provides a basis for risk stratification and genetic counseling.
Die Pylorusstenose beschreibt eine Verengung im Bereich des Magenausganges. Diese kann angeboren oder erworben sein. Sie führt zu einer gestörten Fortleitung des Mageninhalts in den Zwölffingerdarm und somit zu unstillbarem Erbrechen. Die Behandlung besteht in der Regel in einer operativen Korrektur der Engstelle. Die Erkrankung ist bereits mit der Geburt angelegt und kommt familiär gehäuft vor (evtl. erblich). Die Ursachen sind bislang ungeklärt. Zu finden ist die Krankheit vor allem bei West- und Nordeuropäern mit einer Häufigkeit von 1:300, selten bei Asiaten und fast nie bei Afrikanern. Der Erkrankungsgipfel liegt bei drei Wochen nach der Geburt. Die Krankheit tritt besonders bei den erstgeborenen Jungen auf (Verhältnis Jungen : Mädchen: 4-5 : 1). Der Säugling erbricht (nicht gallig) etwa eine halbe Stunde nach der Mahlzeit die Nahrung schwallartig teilweise oder vollständig. Durch die Magenreizung können sich im Erbrochenen Blutfäden befinden. Danach sucht er wieder nach Nahrung. Direkt nach einer Mahlzeit können gesteigerte Magenbewegungen (Peristaltik) auf der Bauchoberfläche im Oberbauch beobachtet werden. Teilweise ist der vergrößerte Pylorus tastbar. Die betroffenen Kinder sind durch die behinderte Nahrungspassage unterernährt, untergewichtig, ständig hungrig und entsprechend unzufrieden. Sie setzen Hungerstühle von geringer Masse in hoher Frequenz ab.
Die Duodenalatresie, auch als Duodenojejunale Atresie
bezeichnet, ist eine angeborene Entwicklungsstörung,
bei der das Lumen des Zwölffingerdarmes nicht
durchgängig ist. Dies ist entweder durch ein Fehlen
eines kurzen oder längeren Darmanteiles (eine echte
Atresie), eine Membran im Darmlumen oder eine
Fehlanlage der Bauchspeicheldrüse bedingt
(ringförmige Fehlanlage der Bauchspeicheldrüse
Pancreas anulare), wobei in diesem Fall meist nur eine
Duodenalobstruktion vorliegt, s. Duodenalstenose. Die
fehlende Durchgängigkeit bewirkt, dass sich vor der
Geburt Magen und Darmanteil vor der Atresie stark
aufweiten und der übrige Darm klein bleibt, da das
geschluckte Fruchtwasser diesen Darmanteil nicht
erreicht. Ferner liegt ein Polyhydramnion vor.
Abdomenübersicht im Liegen Duodenalatresia bei
einem Neugeborenen mit typischen Double-BubbleAntibacterial Envelope to Prevent Cardiac Implantable Device Infection Infections after placement of cardiac implantable electronic devices (CIEDs) are associated with substantial morbidity and mortality. There is limited evidence on prophylactic strategies, other than the use of preoperative antibiotics, to prevent such infections. We conducted a randomized, controlled clinical trial to assess the safety and efficacy of an absorbable, antibiotic-eluting envelope in reducing the incidence of infection associated with CIED implantations. Patients who were undergoing a CIED pocket revision, generator replacement, or system upgrade or an initial implantation of a cardiac resynchronization therapy defibrillator were randomly assigned, in a 1:1 ratio, to receive the envelope or not. Standard-of-care strategies to prevent infection were used in all patients. The primary end point was infection resulting in system extraction or revision, long-term antibiotic therapy with infection recurrence, or death, within 12 months after the CIED implantation procedure. The secondary end point for safety was procedure-related or system- related complications within 12 months. The TYRX envelope is an absorbable single-use envelope designed to hold a CIED when the device is implanted in the body. The envelope is constructed from a multifilament knitted mesh and coated with an absorbable polymer mixed with minocycline and rifampin, which elutes the antibiotics into the local tissue for a minimum of 7 days. The envelope is fully absorbed in approximately 9 weeks.
Procedure Characteristics Procedure characteristics were balanced between the two groups. A total of 3429 patients who were assigned to the control group did not receive the envelope, and 3371 patients who were assigned to the envelope group received the envelope at the time of the CIED procedure. In the envelope group, the envelope was not successfully implanted in 10 procedures owing to limited pocket space; thus, the envelope was successfully implanted in 99.7% of procedure attempts (3371 of 3381 attempts) by 646 implanting physicians. The mean procedure time did not differ significantly between the two groups (56.3±46.2 minutes in the envelope group and 55.0±48.0 minutes in the control group; between-group difference, 1.3 minutes; 95% confidence interval [CI], −0.9 to 3.5).
Kaplan–Meier Curves for First Major CIED Infection. Results are for the overall randomized cohort through 12 months (Panel A) and through all follow-up (Panel B) and were not adjusted for multiple comparisons. Hazard ratios are derived from Cox regressions, with stratification according to device class, and indicate the relative (envelope vs. control) risk of CIED infection. The insets show the same data on an enlarged y axis. Major or minor CIED infections within 12 months occurred in 50 patients in the envelope group and 75 patients in the control group (12-month Kaplan–Meier estimated event rate, 1.5% and 2.2%, respectively; hazard ratio, 0.67; 95% CI, 0.47 to 0.96). Through the entire follow-up period, major CIED infections occurred in 32 patients in the envelope group and 51 patients in the control group (36-month Kaplan–Meier estimated event rate, 1.3% and 1.9%, respectively; hazard ratio, 0.63; 95% CI, 0.40 to 0.98)
Subgroup Analysis of Major CIED Infections through 12 Months. The P values are for the interaction between the randomization group and the subgroup variable. The body-mass index is the weight in kilograms divided by the square of the height in meters. The CIED type is the type planned at randomization: low power (pacemaker or cardiac resynchronization therapy pacemaker) or high power (implantable cardioverter– defibrillator or cardiac resynchronization therapy defibrillator). COPD denotes chronic obstructive pulmonary disease.
Discussion WRAP-IT was a randomized, controlled clinical trial to assess the safety and efficacy of the TYRX absorbable antibiotic-eluting envelope. In a population of patients who were at increased risk for CIED pocket infection, the envelope was significantly more effective at preventing infection than standard infection-control strategies alone. The efficacy objective was met, with 40% fewer patients in the envelope group than in the control group having a major infection through 12 months of follow-up. The envelope was successfully implanted in 99.7% of procedure attempts, and the safety objective was met, because the envelope group did not have a higher incidence of CIED procedure-related or system-related complications than the control group. Some limitations should be considered when interpreting the results of this trial. First, consecutive patients were not enrolled because this trial was limited to patients receiving generators from one device manufacturer (leads from other manufacturers were permitted). Second, the envelope was commercially available at the time of the trial, which may have influenced participation in the trial. Third, the use of immunosuppressive agents was not balanced between the trial groups; however, among these patients, there was only one major infection within the first 12 months, and therefore this is unlikely to influence interpretation of the results. Fourth, the lack of data on antibiotic susceptibility limits our ability to address the unknown risk of the development of antibiotic resistance. Fifth, periprocedure and postprocedure infection-prevention strategies including antibiotic use were not controlled; however, there is not yet clear evidence that a particular strategy influences the infection rate. In conclusion, adjunctive use of an antibacterial envelope resulted in a 40% lower incidence of major CIED infection than standard-of-care infection-prevention strategies alone. Patients who received the envelope did not have more procedure-related or system-related complications than those who did not receive it.
Dabigatran-etexilat ist ein Arzneistoff aus der Gruppe
der Gerinnungshemmer (Antikoagulanzien). Die
Substanz ist ein Prodrug, das nach Umwandlung in das
pharmakologisch aktive Dabigatran den
Blutgerinnungsfaktor IIa (Thrombin) direkt hemmt. Nach
der Einnahme wird der Stoff in der Niere aus dem Blut
gefiltert und über den Urin entsorgt, weshalb
Dabigatranetexilat als Gerinnungshemmer für Patienten
mit Niereninsuffizienz nicht geeignet ist.
Dabigatranetexilat ist für die perorale Verabreichung
geeignet und in dieser Form (Handelsname Pradaxa;
Hersteller Boehringer Ingelheim) in der EU zugelassen
zur Vorbeugung gegen die Bildung von Blutgerinnseln
in den Venen nach elektivem chirurgischen Knie- oder
Hüftgelenksersatz, seit 2011 außerdem zur
Schlaganfallvorbeugung bei Patienten mit
Vorhofflimmern und Schlaganfallrisiko.
Dabigatran bindet kompetitiv und reversibel direkt an
Thrombin und blockiert dessen Wirkung, so dass die
Umwandlung von Fibrinogen zu Fibrin und damit eine
Gerinnselentstehung unterbleibt. Dabigatran verhindert
somit auch die Thrombin-induzierte Verklumpung der
Blutplättchen (Thrombozytenaggregation). Die
gerinnungshemmende (antikoagulatorische) Wirkung
des Dabigatran korreliert mit seiner Konzentration im
Plasma: die therapeutische Konzentration liegt bei
0,05–0,1 mg/l, messbar als 10–20 % der Norm im
neuen F10a/F2a Gerinnungstest EXCA (extrinsic
coagulation activity assay).
PlättchenDabigatran for Prevention of Stroke after Embolic Stroke of Undetermined
Source
Cryptogenic strokes constitute 20 to 30% of
ischemic strokes, and most cryptogenic strokes are
considered to be embolic and of undetermined
source. An earlier randomized trial showed that
rivaroxaban is no more effective than aspirin in
preventing recurrent stroke after a presumed
embolic stroke from an undetermined source.
Whether dabigatran would be effective in
preventing recurrent strokes after this type of
stroke was unclear.
We conducted a multicenter, randomized, double-
blind trial of dabigatran at a dose of 150 mg or 110
mg twice daily as compared with aspirin at a dose
of 100 mg once daily in patients who had had an
embolic stroke of undetermined source. The
primary outcome was recurrent stroke. The primary
safety outcome was major bleeding.
Trial Population
Patients 60 years of age or older were eligible for
enrollment if they had had an embolic stroke of
undetermined source within the previous 3 months
or, if they had at least one vascular risk factor,
within the previous 6 months; patients 18 to 59
years of age were eligible if they had had a
qualifying stroke within the previous 3 months and
had at least one additional vascular risk factorDiscussion The RE-SPECT ESUS trial showed no significant difference between the effect of dabigatran and that of aspirin on the risk of recurrent stroke among patients with embolic stroke of undetermined source. The rate of recurrent stroke was 4.1% per year among patients in the dabigatran group and 4.8% per year among patients in the aspirin group. Dabigatran was associated with major bleeding in 1.7% of the patients per year, and aspirin was associated with major bleeding in 1.4% of the patients per year. The percentages were similar in the two groups in all subcategories of major bleeding, but more patients in the dabigatran group than in the aspirin group had clinically relevant nonmajor bleeding. Our hypothesis was that dabigatran would be more effective than aspirin for stroke prevention in patients with embolic stroke of undetermined source because many of these patients might have had an unrecognized source of cardiac embolism, including atrial fibrillation. Post hoc analysis suggested that dabigatran may have had an effect on stroke recurrence after 1 year, but no inferences can be made because of the post hoc nature of the analysis. A possible explanation for this temporal pattern might be a progressive increase in the occurrence of asymptomatic, undetected atrial fibrillation and other cardiac sources of embolism over time.
Es geht um genomische Kollision
LIM and senescent cell antigen-like-containing domain
protein 1 is a protein that in humans is encoded by
the LIMS1 gene. The protein encoded by this gene is
an adaptor protein which contains five LIM domains, or
double zinc fingers. The protein is likely involved in
integrin signaling through its LIM domain-mediated
interaction with integrin-linked kinase, found in focal
adhesion plaques. It is also thought to act as a bridge
linking integrin-linked kinase to NCK adaptor protein 2,
which is involved in growth factor receptor kinase
signaling pathways. Its localization to the periphery of
spreading cells also suggests that this protein may play LIMS1 mRNA gene expression and cis-eQTL effect of rs893403
a role in integrin-mediated cell adhesion or spreading.
LIMS1 has been shown to interact with Integrin-linked
kinase and NCK2.
GRIP and coiled-coil domain-containing protein 2 is
a protein that in humans is encoded by the GCC2 gene.
The protein encoded by this gene is a
peripheral membrane protein localized to the trans-
Golgi network. It is sensitive to brefeldin A. This
encoded protein contains a GRIP domain which is
thought to be used in targeting. Two alternatively
spliced transcript variants encoding
different isoforms have been described for this gene.
GCC2mRNA gene expression and cis-eQTL effect of
rs893403We used a publicly available catalogue of known copy-number variants generated with the use of 2.1 M NimbleGen comparative genome hybridization arrays. From this data set, a total of 3266 copy-number variants were mapped to the human reference genome hg18 (accessed July 2010). To optimize the power of this study, we selected only copy-number polymorphisms that had a global minor allele frequency of more than 10%, corresponding to the expected homozygosity rates of more than 1%. For the 50 genotyped SNPs, we performed strict quality-control analysis of genotypes that included per-SNP and per- recipient genotyping rates of more than 95%, elimination of monomorphic SNPs, and elimination of markers that significantly deviated from the Hardy–Weinberg equilibrium within each ethnic group. On the basis of the HapMap3 data, we found a single nucleotide polymorphism (SNP) that was an informative tag (r2>0.8) for 50 (57%) of the identified deletions. In the Kaplan–Meier analysis, we observed that a single SNP (rs893403) surpassed a Bonferroni- corrected significance threshold and reached a false discovery rate of 0.3%. Kidney transplant recipients who were homozygous for the deletion- tagging allele had an approximately 84% higher risk of rejection than those who did not have this genotype.
Genomic Mismatch at LIMS1 Locus and Kidney Allograft Rejection In the context of kidney transplantation, genomic incompatibilities between donor and recipient may lead to allosensitization against new antigens. We hypothesized that recessive inheritance of gene-disrupting variants may represent a risk factor for allograft rejection. We performed a two-stage genetic association study of kidney allograft rejection. In the first stage, we performed a recessive association screen of 50 common gene-intersecting deletion polymorphisms in a cohort of kidney transplant recipients. In the second stage, we replicated our findings in three independent cohorts of donor–recipient pairs. We defined genomic collision as a specific donor– recipient genotype combination in which a recipient who was homozygous for a gene- intersecting deletion received a transplant from a nonhomozygous donor. Identification of alloantibodies was performed with the use of protein arrays, enzyme-linked immunosorbent assays, and Western blot analyses. In the discovery cohort, which included 705 recipients, we found a significant association with allograft rejection at the LIMS1 locus represented by rs893403 (hazard ratio with the risk genotype vs. nonrisk genotypes, 1.84; 95% confidence interval. We found that the LIMS1 locus appeared to encode a minor histocompatibility antigen. Genomic collision at this locus was associated with rejection of the kidney allograft and with production of anti-LIMS1 IgG2 and IgG3.
Discovery Phase. Panel A shows our strategy for selecting high-priority deletions for tagging and typing in the discovery cohort. A total of 44 of 50 deletions were successfully tagged and genotyped in the discovery cohort; 6 of 50 deletion-tagging single-nucleotide polymorphisms (SNPs) were either monomorphic or failed our genotype quality-control analysis. Annotations were based on the human reference genome hg18 (accessed in July 2010). Copy-number polymorphisms (CNPs) were common copy-number variants (CNVs with an allele frequency of >1%). MAF denotes minor allele frequency. Panel B shows the probability–probability plot for the genetic screen for rejection in the discovery cohort of 705 recipients under a recessive model. The blue dots represent P values for 44 successfully typed common deletions; the red dotted lines represent significance thresholds of 0.05 (unadjusted analysis) and 0.0011 (Bonferroni-corrected for 44 independent tests). The blue dotted line indicates the expectation under the null hypothesis, and the shaded area corresponds to a 95% confidence interval for the null hypothesis of no association. The top SNP (rs893403) represents a near-perfect tag (r2=0.98) for a common 1.5-kb deletion (CNVR915.1) on chromosome 2q12.3. Panel C shows the genomic characteristics of the 44 CNP- tagging SNPs that were tested in the discovery phase. Plus–minus values are means ±SD.
Effects of rs893403 on Rejection-free Allograft Survival in Study Cohorts. Panel A shows the results in the discovery phase (involving 705 kidney transplant recipients [the Columbia cohort] who had either a nonrisk genotype [blue] or a risk genotype [red]). Tick marks indicate censored data. Panel B shows the results in the replication phase, which involved a stratified analysis of three other cohorts (Belfast, TransplantLines, and Torino) that included a total of 2004 donor– recipient pairs. The P values correspond to the minimally adjusted model, with adjustment for cohort only (if applicable). Panel C shows the results in all the cohorts combined, which involved a stratified analysis of the four cohorts (i.e., 2709 kidney transplants [in 705 recipients from the discovery cohort and 2004 donor–recipient pairs from the replication cohorts]). Panel D shows the estimated hazard ratios (with 95% confidence intervals) of rejection in each of the four cohorts individually, in all the replication cohorts, and in all the cohorts combined. The effects were estimated before (blue [recipient only]) and after (red [donor– recipient pairs]) accounting for donor compatibility in order to show that the inclusion of genetic information from the donors resulted in consistently improved hazard ratio estimates.
Detection of Anti-LIMS1 Antibodies in Kidney Transplant Recipients at Genetic Risk for Rejection. Panel A shows the change in intensity (x axis) as compared with the −log P value (y axis) for the top-ranking proteins on the basis of the mean signal intensity in a protein array; the change is calculated as a ratio of the mean normalized intensity in the high-risk rejection group to the mean normalized intensity of all other groups (termed “fold change”). The findings suggest the presence of anti-LIMS1 reactivity in high-risk recipients with rejection. Panel B shows the normalized intensity levels for LIMS1 on the protein array for the comparison between the high-risk rejection group and all other groups (P=0.002); the horizontal lines represent the group means. Panel C shows the results of anti-LIMS1 total IgG seroreactivity studies with the use of an enzyme-linked immunosorbent assay that were performed in 318 persons across seven genotype- and phenotype-discordant groups. The results are shown as the change in the optical density (OD), defined as a ratio of the measured OD for each sample to the mean OD of the same 5 normalization controls (serum samples obtained from healthy persons) that were used on each plate. These studies included 52 controls who had not undergone transplantation (Control), 37 recipients who were homozygous for the risk allele and did not have rejection (Risk-NR), 31 recipients who were homozygous for the risk allele and had rejection (Risk-R; in red), 50 recipients who were heterozygous for the risk allele and did not have rejection (Het-NR), 50 recipients who were heterozygous for the risk allele and had rejection (Het-R), 63 recipients who were homozygous for the non-risk– associated allele and did not have rejection (Hom-NR), and 35 recipients who were homozygous for the non-risk– associated allele and had rejection (Hom-R). Total IgG seroreactivity was detected only in recipients with a high- risk genotype who had rejection.
Discussion In this study, we examined a genomic-collision scenario in which an allograft recipient was homozygous for a deletion polymorphism and received a kidney allograft from a donor who had at least one normal allele. In the analysis of four large kidney transplant cohorts, we found that the genomic collision at chromosome 2q12.3 led to a risk of rejection that was nearly 60% higher than the risk among donor–recipient pairs with noncollision genotypes. The risk associated with the collision genotype is equivalent to a mismatch of three of six HLA alleles, which is both clinically significant and potentially modifiable by genetic testing and matching. The genomic collision at chromosome 2q12.3 would be expected to occur in approximately 12 to 15% of transplants from unrelated donors among persons of European and African ancestry but would be very rare among persons of East Asian ancestry. Discussion In this study, we examined a genomic-collision scenario in which an allograft recipient was homozygous for a deletion polymorphism and received a kidney allograft from a donor who had at least one normal allele. In the analysis of four large kidney transplant cohorts, we found that the genomic collision at chromosome 2q12.3 led to a risk of rejection that was nearly 60% higher than the risk among donor–recipient pairs with noncollision genotypes. The risk associated with the collision genotype is equivalent to a mismatch of three of six HLA alleles, which is both clinically significant and potentially modifiable by genetic testing and matching. The genomic collision at chromosome 2q12.3 would be expected to occur in approximately 12 to 15% of transplants from unrelated donors among persons of European and African ancestry but would be very rare among persons of East Asian ancestry. Taken together, our results provide support for genomic collision at chromosome 2q12.3 contributing to the risk of allograft rejection and point to LIMS1 as a potential minor histocompatibility antigen encoded by this locus. In addition, we found that the LIMS1 protein was expressed in other commonly transplanted tissues, such as the heart and lung, but follow-up studies will be useful in determining whether our findings are generalizable to other organs.
Phosphoinositid-3-Kinasen, kurz PI3K, sind Enzyme aus der Gruppe der Transferasen, die in sämtlichen eukaryotischen Zellen auffindbar sind. Sie gehören nach der EC-Klassifikation zur Klasse 2.7. Die PI3-Kinase ist für die Bildung von Phosphatidylinositol-3,4,5-trisphosphat (PIP3) aus Phosphatidylinositol-4,5- bisphosphat PIP2 zuständig. Das PIP3 ist u.a. für die Aktivierung einer weiteren Kinase, der Proteinkinase B (PKB) verantwortlich. Die PKB fördert das Zellwachstum und verhindert v.a. die Apoptose. Außerdem reguliert sie auch metabolische Reaktionen. Phosphoinositid-3-Kinasen katalysieren einen Phosphorylierungsvorgang der 3'-OH Position am Inositolring von einigen Phospholipiden (Phosphatidylinositole). Diese dienen als Andockstelle für weitere Proteine (z.B. Proteinkinase B), sodass sie einen wichtigen Signalweg, den PI3K/Akt-Signalweg, aktivieren. Phosphoinositid- 3-Kinasen sind in eine Vielzahl von zellulären Schlüsselfunktionen wie etwa Zellwachstum, Proliferation, Migration, Differenzierung, Überleben (Apoptose über PKB) und Zelladhäsion involviert. Fehlfunktionen können die Entstehung von Krankheiten (z.B. Allergien, Herzkrankheiten, Entzündungsprozesse oder Krebs) begünstigen.
Was ist PIK3CA?
The phosphatidylinositol-4,5-bisphosphate 3-kinase,
catalytic subunit alpha (the HUGO-approved official
symbol = PIK3CA; HGNC ID, HGNC:8975), also called
p110α protein, is a class I PI 3-kinase catalytic subunit.
The human p110α protein is encoded by
the PIK3CAgene.
Phosphatidylinositol-4,5-bisphosphate 3-kinase (also
called phosphatidylinositol 3-kinase (PI3K)) is
composed of an 85 kDa regulatory subunit and a 110
kDa catalytic subunit. The protein encoded by this gene
represents the catalytic subunit, which uses ATP to
phosphorylate phosphatidylinositols (PtdIns), PtdIns4P
and PtdIns(4,5)P2. The involvement of p110α in human
cancer has been hypothesized since 1995. Support for
this hypothesis came from genetic and functional
studies, including the discovery of common activating Bestimmung des PIK3CA-Mutations-Status
PIK3CA missense mutations in common human tumors. Die Bestimmung des PIK3CA-Mutations-
PIK3CA mutations are present in over one-third of Status ist u. a. wichtig bei der Behandlung
breast cancers, with enrichment in the luminal and in von Patienten mit kolorektalem Karzinom
human epidermal growth factor receptor 2-positive (CRC), Mammakarzinom und
subtypes (HER2 +). The three hotspot mutation Lungenkarzinom. Hierbei wird untersucht,
positions (GLU542, GLU545, and HIS1047) have been ob es im PIK3CA-Gen (Abk. für
widely reported till date. PIK3CA participates in a „Phosphatidylinositol-4,5-Bisphosphate 3-
complex interaction within the tumor Kinase Catalytic Subunit Alpha“) im Laufe
microenvironment in this phenomenon. der Entstehung des Tumors zu onkogenen
Mutationen gekommen ist oder nicht.Alpelisib for PIK3CA-Mutated, Hormone Receptor–Positive Advanced Breast Cancer PIK3CA mutations occur in approximately 40% of patients with hormone receptor (HR)–positive, human epidermal growth factor receptor 2 (HER2)–negative breast cancer. The PI3Kα-specific inhibitor alpelisib has shown antitumor activity in early studies. In a randomized, phase 3 trial, we compared alpelisib (at a dose of 300 mg per day) plus fulvestrant (at a dose of 500 mg every 28 days and once on day 15) with placebo plus fulvestrant in patients with HR- positive, HER2-negative advanced breast cancer who had received endocrine therapy previously. Patients were enrolled into two cohorts on the basis of tumor-tissue PIK3CA mutation status. The primary end point was progression- free survival, as assessed by the investigator, in the cohort with PIK3CA-mutated cancer; progression-free survival was also analyzed in the cohort without PIK3CA-mutated cancer. Secondary end points included overall response and safety.
Kaplan–Meier Analysis of Progression-free Survival. In the cohort of patients with PIK3CA- mutated cancer, the median progression-free survival was 11.0 months in the alpelisib–fulvestrant group and 5.7 months in the placebo–fulvestrant group (Panel A). The primary end point crossed the prespecified Haybittle–Peto boundary (one-sided P≤0.0199) (Panel A). Symbols indicate censored data. In the cohort without PIK3CA-mutated cancer, the median progression-free survival was 7.4 months in the alpelisib–fulvestrant group and 5.6 months in the placebo–fulvestrant group (Panel B). The gene PIK3CA encodes for the alpha isoform of phosphatidylinositol 3-kinase (PI3Kα).
Subgroup Analysis of Progression-free Survival in the Cohort with PIK3CA- Mutated Cancer. Confidence intervals have not been adjusted for multiplicity. Inferences drawn from the confidence intervals may not be reproducible. The previous chemotherapy subgroup was based on the last line of chemotherapy received. Patients may have received chemotherapy in the context of both neoadjuvant and adjuvant therapy. Patients may have had more than one PIK3CA mutation. There were multiple subtypes of E545 and H1047 mutations. CDK denotes cyclin-dependent kinase.
Proof-of-concept criteria were not met in the cohort of patients without PIK3CA-mutated cancer at the final efficacy analysis. The median progression-free survival was 7.4 months (95% CI, 5.4 to 9.3) in the alpelisib– fulvestrant group and 5.6 months (95% CI, 3.9 to 9.1) in the placebo–fulvestrant group (hazard ratio for progression or death, 0.85; 95% CI, 0.58 to 1.25; posterior probability of true hazard ratio
Safety The total safety population included 284 patients who received alpelisib– fulvestrant and 287 who received placebo–fulvestrant. The adverse events of any grade that occurred in at least 35% of the patients in either group were hyperglycemia (in 63.7% of the patients who received alpelisib–fulvestrant and 9.8% of those who received placebo– fulvestrant), diarrhea (in 57.7% and 15.7%, respectively), nausea (in 44.7% and 22.3%), decreased appetite (in 35.6% and 10.5%), and rash (in 35.6% and 5.9%) or maculopapular rash (in 14.1% and 1.7%)
Discussion These results show improvements in patients’ outcomes with the addition of an α-specific PI3K inhibitor to standard treatment for PIK3CA-mutated, HR-positive, HER2-negative advanced breast cancer, findings that validate PIK3CA as an important treatment target in this population. Patients with PIK3CA-mutated, HR-positive, HER2-negative advanced breast cancer that had progressed during or after the receipt of endocrine therapy had significantly longer progression-free survival when they received alpelisib–fulvestrant than when they received placebo–fulvestrant, with an estimated 35% lower risk of progression or death. A clinically relevant treatment benefit was not observed for alpelisib–fulvestrant in the cohort without PIK3CA-mutated cancer. In the cohort with PIK3CA-mutated cancer, alpelisib– fulvestrant was also associated with significantly higher percentages of patients with tumor response than was placebo–fulvestrant, a finding that is consistent with observations from the phase 1b study. Progression-free survival was similar in the placebo groups in the two cohorts defined according to PIK3CA mutation status. Alpelisib has activity in patients with PIK3CA-mutated, HR-positive, HER2-negative advanced breast cancer that has progressed during or after treatment with an aromatase inhibitor. Therefore, the integration of genomic testing for PIK3CA mutation into routine clinical practice may be useful in the selection of therapy; validated diagnostic testing procedures are not yet available. This trial shows that treatment with alpelisib–fulvestrant can provide an extension of progression-free survival among patients with PIK3CA-mutated disease. This effect was observed across various subgroups. Preliminary analysis of progression-free survival on the basis of ctDNA results shows a similar effect. In conclusion, this phase 3 trial showed a significant prolongation of progression-free survival and greater overall response with alpelisib–fulvestrant than with placebo–fulvestrant among patients with PIK3CA-mutated, HR-positive, HER2-negative advanced breast cancer who had disease that had relapsed or progressed during or after the receipt of previous endocrine therapy.
Muco-Obstructive Lung Diseases A spectrum of lung diseases that affect the airways, including chronic obstructive pulmonary disease (COPD), cystic fibrosis, primary ciliary dyskinesia, and non–cystic fibrosis bronchiectasis, can be characterized as muco-obstructive diseases. These diseases have the clinical features of cough, sputum production, and episodic exacerbations that are often associated with a diagnosis of chronic bronchitis. However, neither “chronic bronchitis” nor “hypersecretory diseases” adequately describes the diffuse mucus obstruction, airway-wall ectasia, chronic inflammation, and bacterial infection that are typical of these conditions; therefore, “muco-obstructive” may be a preferred descriptive term. Although asthma can also be associated with diffuse airway mucus obstruction, its distinct pathophysiological mechanisms preclude discussion in this grouping. In healthy persons, a well-hydrated mucus layer is transported rapidly (at a rate of approximately 50 μm per second) from the distal airways toward the trachea. In muco-obstructive diseases, epithelial defects in ion–fluid transport, mucin secretion, or a combination of these lead to hyperconcentrated (dehydrated) mucus, failed mucus transport, and mucus adhesion to airway surfaces. Mucus that is accumulated in the trachea can be expectorated by cough as phlegm or sputum. Mucus in the small airways cannot be cleared by cough and accumulates, forming the nidus for airflow obstruction, infection, and inflammation. Biochemical and Biophysical Properties of Mucus Relative to Airway Function Human airway mucus is a hydrogel composed of approximately 98% water, 0.9% salt, 0.8% globular proteins, and 0.3% high-molecular-weight mucin polymers.12 The hydration (concentration) status of mucus is measured as the wet-to-dry content of mucus (i.e., the percentage of a given volume that consists of nonvolatile solids) or as the mucin concentration determined by light-scattering techniques.The correlation between the two measurements (in healthy persons or those with muco-obstructive disease) is high, which allows both measures to be used to describe this mucus property. The two major synthesized and secreted respiratory mucins, MUC5B and MUC5AC, are physically very large, spanning 0.2 to 10 μm in length for single polymers. The secreted mucin polymers interweave to form mesh-like gels with mesh sizes that are concentration dependent (i.e., higher concentrations are associated with smaller mesh sizes). MUC5B and MUC5AC share many features, including their multimeric organization and high carbohydrate content (approximately 75% of total weight).
Muco-Obstructive Disease Pathogenesis, Mucin Species, and Total Mucin Concentrations in Health and Disease. Panel A shows the progression from normal to muco- obstructed airways. In healthy persons (left), well-balanced epithelial sodium (Na+) absorption and secretion of chloride anions (Cl−) hydrates airway surfaces and promotes efficient mucociliary clearance (MCC). In persons with muco- obstructive lung disease (middle), an imbalance of ion transport coupled with mucin hypersecretion increases mucin concentrations in the mucus layer, osmotically compresses the periciliary layer (PCL), and abolishes MCC. The adherent mucus may be expelled as sputum by cough (upper right). Mucus that cannot be expelled by cough accumulates, concentrates, obstructs airflow, and becomes the nidus for infection (lower right). CFTR denotes cystic fibrosis transmembrane conductance regulator, and ENaC the epithelial sodium channel. Panel B shows domain structures and relative sizes of the secreted mucins (MUC5AC and MUC5B) and tethered mucins (MUC1, MUC4, and MUC16). For reference, the globular protein albumin (ALB) is shown. The secreted mucins are composed of monomers with N terminals (MUC5AC, green; MUC5B, light blue), glycosylated domains, and C terminals (MUC5AC, yellow; MUC5B, purple). Both the C–C terminal dimers and N–N terminal multimers are linked by S–S bonds. The glycosylated domains contain sugar side chains from 2 to 15 sugar molecules in length, often terminally capped with sialic acid or sulfate, which gives mucins a negative charge. The glycosylation domains provide mucin hydration, reflecting the avidity of sugar molecules for water, and a combinatorial library of binding sites capable of trapping most inhaled materials with a low but sufficient binding affinity to mediate clearance. The tethered mucins have cytoplasmic N-terminal (dark gray), transmembrane (light gray), and heavily glycosylated extracellular domains.
Two-Gel Model of Mucus Transport. Panel A, left, shows the classic “gel-on-liquid” model showing a mucus layer (MUC5AC and MUC5B) and a “sol” layer as liquid surrounding cilia. Panel A, right, shows the “two-gel” model in which the mucus layer remains the same, but a periciliary layer (gel) is formed by tethered macromolecules, including MUC1, MUC4, and MUC16. Panel B shows epithelial ion and water transport in normal human airway epithelia. The percentage of solids in the normal mucus layer and osmotic pressures (π) of the normal mucus layer (πML) and PCL (πPCL) in pascals (Pa) are noted. An ENaC in the apical membrane mediates sodium and liquid absorption.21 In parallel, the epithelium secretes chloride and bicarbonate anions through CFTR and calcium-activated chloride channels (CaCC). The balance between active sodium absorption and secretion of chloride and bicarbonate anions, and hence fluid flow, is regulated in part by lumenal concentrations of extracellular ATP, interacting through P2Y2 receptors, and adenosine (ADO), interacting through A2b receptors. Panel C describes the relative water-drawing powers of the mucus layer (πML) and PCL (πPCL) interfaced to epithelial cell–mediated fluid absorption or secretion (blue arrows). The length of the Hookean springs (to the right of the blue arrows) denotes the height of the PCL (purple) or mucus layer (green), and the spring diameter is inversely proportional to osmotic pressure. In a normal state (left), πML is lower than πPCL, represented by a green spring (πML) with a diameter larger than the purple spring (πPCL). In a dehydrated state (right), persistent or abnormal absorption initially removes fluid from the lower-osmotic-pressure mucus layer but ultimately coordinately removes fluid from both the mucus layer and the PCL. The osmotic pressures of both layers are increased and equalized (smaller spring diameters) and volume depleted (springs shortened). This state osmotically compresses the cilia and produces mucus stasis. Panel D shows the relationship between mucus concentration (i.e., percent solids) and osmotic pressure (Pa).
Muco-Inflammatory Positive-Feedback (“Vicious”) Cycles in Muco-Obstructive Disease. Panel A shows the basal muco-obstructive disease state in which surface hydration and mucus concentration may be relatively normal but vulnerable to muco-obstruction with insults (e.g., aspiration or viral infection). Panel B shows insult-triggered formation of abnormal mucus plaque. Insults increase mucin secretion not accompanied by adequate hydration owing to abnormal fluid-secretory responses. Hyperconcentrated mucus activates resident macrophages (pink) and produces hypoxia in subjacent airway epithelia (blue). Panel C shows the generation of persistent, muco-inflammatory positive-feedback cycles. Activated resident macrophages release interleukin-1β (left), and hypoxic necrotic epithelia (blue) release interleukin-1α (right). Interleukin-1α and -1β activate epithelial interleukin-1 receptors (IL1R1) to induce mucin biosynthesis mediated by SPDEF and ERN2 and expression of proinflammatory cytokines and chemokines (e.g., IL8 and CXCL1). Accelerated mucin secretion without proper hydration worsens mucus hyperconcentration on airway surfaces and stimulates hypoxic macrophages and epithelia to release additional interleukin-1α and -1β in positive-feedback loops (denoted by a plus sign). Secretion of proinflammatory mediators induces parallel neutrophil-mediated inflammation; parallel neutrophil-mediated, protease-induced mucin secretion; and other positive-feedback cycles (not shown). Po2 denotes partial pressure of oxygen.
Diagnosis Disease-specific criteria assist in the diagnosis of each muco-obstructive lung disease: for cystic fibrosis, levels of chloride anions in sweat and genetic testing; for COPD, exposure history and spirometry; for primary ciliary dyskinesia, nasal nitric-oxide measurements, cilia waveform analyses, and genetic testing; and for non–cystic fibrosis bronchiectasis, CT scanning. The tools to make a general diagnosis of muco-obstructive disease are also available. Therapies for Muco-Obstructive Diseases Disease-specific therapies for one muco-obstructive disease, cystic fibrosis, are now available. Ivacaftor (VX- 770) is a potentiator of residual CFTR function that has been approved for patients with cystic fibrosis with gating and some splicing CFTR mutations. Ivacaftor provided a model for the development of therapies for muco-obstruction, as evidenced by impressive associations between improved airway-surface hydration in vitro, peripheral and central mucociliary clearance in vivo, and clinically relevant outcomes (e.g., forced expiratory volume in 1 second [FEV1]). Hydrators Perhaps the most direct approach in the treatment of muco-obstructive lung diseases is to reduce the concentration of pathologic mucus — that is, rehydrate it. The currently available approach to achieve this goal is inhalation of osmotically active aerosols (e.g., hypertonic saline or mannitol). In clinical development are modulators of ion transport that may redirect airway epithelial ion transport from net absorptive to secretory directions, providing a mechanism for epithelial restoration of airway-surface hydration. Mucolytics Two recent observations suggest that attacking abnormal mesh and gel properties of mucus in addition to reducing concentration may be therapeutic. First, mucus viscosity is a key variable governing the mucus cohesive and adhesive properties relevant to cough efficiency, and reductions in viscosity with mucin S–S bond–reducing or surfactant agents decrease mucus adhesion and cohesion independent of concentration. Second, the mucus plaques or flakes that are recovered from young patients with cystic fibrosis during bronchoalveolar lavage are unable to swell and dissolve in excess solvent. Inhaled acetylcysteine has not proved to have the mucin-reductive activity required for a therapeutic effect. Conclusions Muco-obstructive diseases are characterized by mucus hyperconcentration. The four muco-obstructive diseases differ with respect to the epithelial abnormalities that produce mucus hyperconcentration but follow a final common path of mucus concentration–dependent formation of mucus plaques and plugs.
A 60-year-old man presented to the otorhinolaryngology
clinic with a feeling of nasal obstruction and postnasal
drip that had developed 10 years earlier and had
worsened over the past month. He also had a sensation
of a foreign body in his throat. Symptoms worsened
when he was lying flat. On physical examination, no
deviation of the nasal septum or hypertrophy of the
inferior turbinates was detected. Nasopharyngoscopy
revealed a smooth, pink, cystic mass in the midline of
his nasopharynx, with no obstruction of the openings to
the eustachian tubes (the image shows the view
through the right naris). Computed tomography of the
head revealed a 2.5 cm by 1.3 cm by 1.4 cm cystic
tumor in the midline of the nasopharynx without
intracranial extension. The findings were consistent with
Tornwaldt’s cyst, a benign cyst that arises between the
roof of the nasopharynx and the remnant of the
notochord. Because the patient was symptomatic, he
underwent surgical marsupialization of the cyst. On
follow-up examination 3 months after surgery, he no
longer had the sensation of a foreign body in his throat,
his nasal symptoms were reduced, and there was no
recurrence of the cyst. Eine Tornwaldt-Zyste (lat.: Bursa pharyngea(lis)),
(gelegentlich auch in den Schreibweisen Thornwaldt- oder
Thornwald-Zyste) ist eine benigne Zyste (eine gutartige,
flüssigkeitsgefüllte Raumforderung), die im oberen hinteren
Nasopharynx (Nasen-Rachenraum) lokalisiert ist.
Sie wird meist zufällig bei der Computertomografie (CT) oder
Magnetresonanztomografie (MRT) des Kopfes als in der
Mittellinie gelegene, gut abgegrenzte rundliche
Raumforderung diagnostiziert.A 55-Year-Old Man with Jaundice A 55-year-old man with a history of opioid use disorder and hepatitis C virus (HCV) infection presented to this hospital with jaundice. Four months before the current presentation, the patient was released from prison after a 2-year incarceration. After he left prison, he resumed injecting heroin and had three episodes of overdose. He was evaluated at another hospital for symptoms of depression and was admitted for psychiatric treatment. During that admission, sublingual buprenorphine–naloxone therapy was initiated, and the patient was discharged. One day after discharge and 5 weeks before the current presentation, headache, body aches, sweats, diarrhea, and nausea developed. The patient presented to a clinic for substance use disorder that is affiliated with the other hospital and reported that he had been unable to obtain sublingual buprenorphine–naloxone from a pharmacy after discharge. The temperature was 36.6°C, the pulse 70 beats per minute, and the blood pressure 100/68 mm Hg. The weight was 72 kg. He appeared to be restless, but the remainder of the physical examination was normal. Urine toxicologic screening was positive for buprenorphine, norbuprenorphine, and norfentanyl; buprenorphine– naloxone therapy was resumed. Three weeks before the current presentation, dark urine and light-headedness developed and did not improve with increased fluid consumption. The patient also noticed slow thinking and arthralgias in the hands, wrists, and elbows. He was evaluated by a new primary care provider.
Limited ultrasonography of the right upper quadrant revealed no bile-duct dilatation, a patent main portal vein with hepatopetal flow, and normal hepatic parenchymal echotexture. Diffuse, hypoechoic gallbladder-wall thickening was present, without gallbladder distention, cholelithiasis, or pericholecystic fluid. Murphy’s sign was negative. There was trace perihepatic ascites. Abdominal Ultrasound Images. Grayscale images (Panels A, B, E, and F) and color Doppler images (Panels C and D) of the right upper quadrant show no intrahepatic or extrahepatic bile-duct dilatation (Panels A and B), a patent main portal vein with flow in the correct direction (Panel C, arrow), trace perihepatic ascites (Panel D, arrows), and a collapsed gallbladder with diffuse, hypoechoic wall thickening (Panels E and F, plus signs). FF denotes free fluid.
A long-held dogma regarding the differential diagnosis of severe acute liver injury is that most cases are due to vascular, viral, or toxic causes. A recent multicenter study examined the causes of severe acute liver injury in patients with an aspartate aminotransferase or alanine aminotransferase level of more than 1000 U per liter and confirmed the leading causes to be ischemic hepatitis, pancreaticobiliary disease, drug-induced liver injury, and viral hepatitis. Ischemic Hepatitis Does this patient have a vascular or ischemic process that is causing acute liver injury? Ischemic hepatitis is defined as a decrease in hepatic blood flow that results in hepatocyte death and necrosis. This condition is commonly seen in critically ill patients, and in many cases, a clear hypotensive episode is not documented, which suggests that the insulting event may be fleeting or subclinical. Recent data further suggest that elevated central venous pressure and cardiac pressures on the right side are important predisposing factors. Pancreaticobiliary Disease The absence of bile-duct dilatation, gallstones, and pancreatic abnormality on ultrasonography provides evidence against a pancreaticobiliary cause of this patient’s liver injury. The magnitude of hyperbilirubinemia and the kinetics of elevation in aminotransferase levels are also not suggestive of biliary disease; the alanine aminotransferase level tends to rise and fall rapidly, and it is uncommon to have a peak bilirubin level of more than 10 mg per deciliter (171 μmol per liter). Drug-Induced Liver Injury Drug-induced liver injury must be considered in all cases of severe acute liver injury or failure; studies have determined it to be the most common cause of acute liver failure in the United States and most of Europe. In this case, the only new medication was buprenorphine–naloxone, which is an exceedingly rare cause of drug-induced liver injury. One report identified buprenorphine–naloxone as the cause of severe liver injury in two patients with chronic HCV infection; the drug had been injected at high doses, and symptomatic hepatitis had occurred within 4 days after the injection. Viral Hepatitis This patient’s clinical presentation is strongly suggestive of acute viral hepatitis. The acute viral infections that most commonly cause severe liver injury are HCV infection (in 8.0% of cases) and hepatitis B virus (HBV) infection (in 2.1% of cases). Hepatitis A virus (HAV), hepatitis delta virus (HDV), Epstein–Barr virus, and cytomegalovirus infections are much less common causes (in 0.3% of cases combined).
Acute HCV Infection Symptoms associated with acute HCV infection occur approximately 9 weeks after exposure. There is a very low level of viremia in the first 2 months after infection, followed by a brisk and exponential increase in viremia over a period of 8 to 10 days. In the subsequent phase, the host immune system responds to the viremia: the aminotransferase levels rise, the HCV RNA viral load plateaus, and symptoms develop. Could this patient have acute HCV infection? Although he reportedly had a history of HCV infection, we have not seen an HCV viral load obtained before this presentation. A positive test for HCV antibodies alone suggests previous exposure but does not confirm a chronic infection. An estimated 25% of persons with exposure to HCV have successful viral clearance; female sex, symptomatic infection, and favorable host genetic factors are associated with spontaneous clearance. Acute HBV Infection The clinical presentation and outcome of acute HBV infection are strongly influenced by the patient’s age; in neonates, viral clearance is exceedingly rare, occurring in less than 5% of cases, whereas in adults, the infection is successfully controlled in more than 95% of cases. The virus itself is noncytopathic, and the symptoms and liver injury are consequences of the host immune response. Liver failure is rare but occurs in approximately 1% of cases. Even in the absence of liver failure, liver injury can be severe; in one case series, the mean alanine aminotransferase level was 1419 U per liter, and the mean bilirubin level 6.5 mg per deciliter (111 μmol per liter). Acute HDV Infection What other factors could explain this patient’s severe acute liver injury? HDV infection is dependent on and exacerbates HBV infection. There are two clinical patterns of infection with HDV: coinfection, in which exposure to HDV and to HBV are simultaneous; and superinfection, in which acute HDV infection occurs in a person with established chronic HBV infection. In cases of coinfection, HDV infection relies on the successful development of HBV infection, which is uncommon in adults, and thus chronic coinfection is rare. HAV Infection HAV infection is a classic cause of severe acute viral hepatitis. Similar to HBV infection, HAV infection has a clinical presentation that varies according to the patient’s age; children are often asymptomatic, whereas more than 70% of adults have symptoms, such as fever, malaise, nausea, vomiting, and abdominal discomfort, that last 2 to 8 weeks.
Sie können auch lesen